Public health touches all aspects of our lives, not just during a pandemic. Thanks to your feedback, this newsletter will continue with COVID updates and address other public health topics, too. To choose what topics land in your inbox, click HERE.
On Friday, two federal judges in Washington and Texas handed down opposing rulings regarding abortion pills. How this plays out could have a huge impact on public health.
I partnered with Dr. Heidi Moseson, a reproductive epidemiologist, and Professors Rachel Rebouche and David Cohen, reproductive health scholars, to translate what’s going on, the public health science in one case (and lack thereof in the other), and what it means for people today.
What’s going on?
Medication abortions use a combination of two drugs: mifepristone (let’s call this “mife”) followed by misoprostol (let’s call this “miso”).
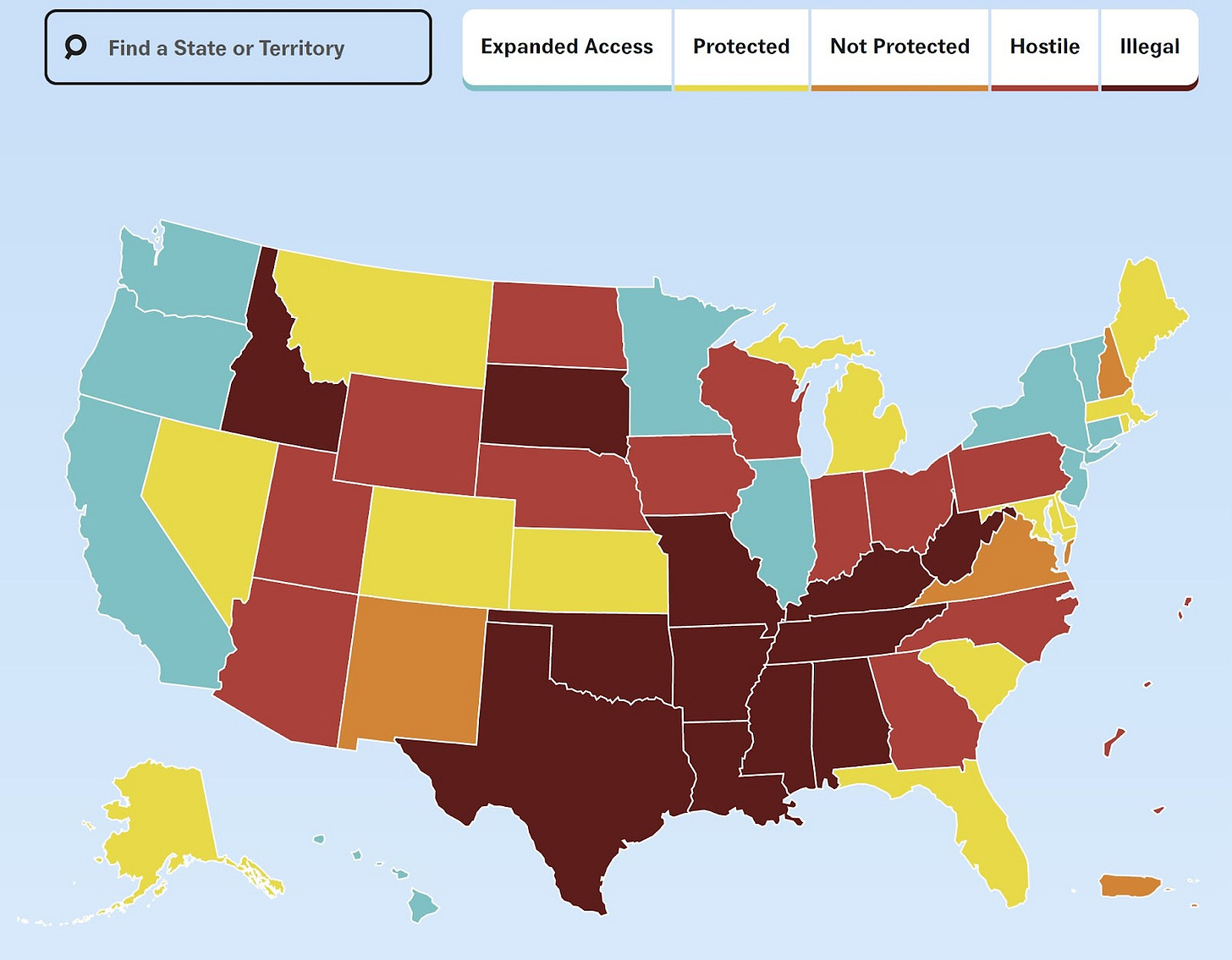
One in two abortions (~490,000) in the U.S. are medication abortions, and this proportion has steadily increased over the years. One reason is that medication abortions can be accessed remotely via mail—salient for the nearly 60% of Americans who now live in a state with limited access to abortion clinics.
Mife and/or miso are also used as a first line of treatment in other life-saving events like miscarriage management, labor induction, and prevention of postpartum hemorrhage.
Last Friday, two opposing legal cases were ruled on medication abortion in the U.S.
Case #1: Texas
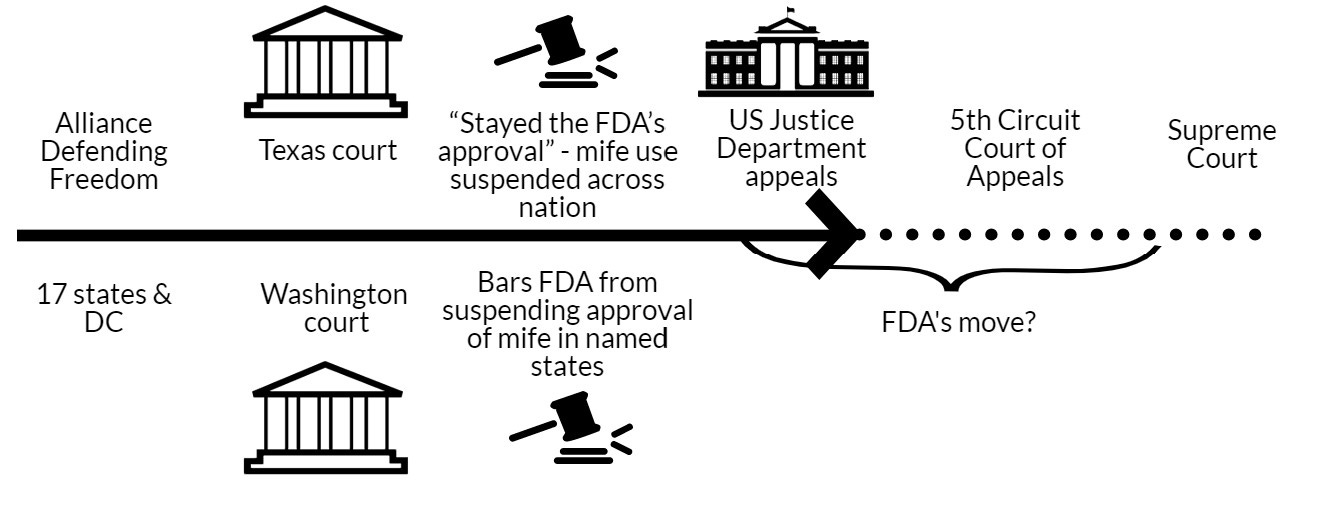
Last November, a group filed a lawsuit with the goal of overturning the FDA’s decades-old approval of mife.
Their lawsuit put forward three claims:
The FDA improperly used its authority to approve mife in 2000. The FDA approved mife under a certain part of the law which says new drugs are approved for “treating serious or life-threatening illnesses.” The legal group argued that pregnancy is not an illness and, therefore, the FDA cannot approve mife.
Mife is not safe. Some studies show that medication abortions with mife have a slightly higher complication rate than procedural abortion, thus the FDA acted improperly when it modified its regulation of mife in 2016, 2019, and 2021.
Mailing abortion pills is illegal. A 150-year-old law (the Comstock Act) bans mailing materials intended for producing abortion, in this case mife for abortion. This law historically only applies for materials that the sender knows would be used for “unlawful” abortions, which is an intent difficult to prove.
The Texas judge could have ruled a gazillion ways: ruled on one of these claims, all of these claims, or anything in between.
Ultimately, the judge “stayed the FDA’s approval”—temporarily put the approval of mife on hold. Suspension of the FDA’s approval would apply to all states (not just in Texas). However, in doing so, he was going to wait for seven days, giving parties time to appeal. What happens on day 8? It’s not clear and may depend on the eye of the beholder (see more below). Regardless, this is unprecedented, as it’s the first time that a court suspended an FDA approval.
As expected, the Justice Department appealed, asking the 5th Circuit Court of Appeals to hold off on the judge’s order until an appeals decision is reached.
Case #2: Washington
In March 2023, 17 states and DC filed a case in Washington state to force the FDA to drop restrictions on mife. This was essentially the exact opposite of the case in Texas.
Their lawsuit put forward two claims:
Mife is safe and effective. The FDA followed all appropriate protocols in its initial approval of mife. Mife’s safety profile is unassailable, and removing this drug from the market would cause “immediate and irreparable injury.”
FDA-imposed restrictions on mife’s use are burdensome and unnecessary. In January 2023, the FDA added a certification requirement for any pharmacy hoping to distribute mife, and kept in place other burdensome forms. These requirements make mife harder to access for patients and are unnecessary.
The Washington judge did not rule on mife restrictions, but instead the judge ordered the FDA not to take any action to change the current situation in which mife is approved. This ruling applies only to the 17 states and DC named in the suit, not nationally.
The public health science (or lack thereof)
One of the questions posed by case #1 —“Is pregnancy an illness?” — is a question asked and answered before. So much so that the scientific evidence has been reviewed, and reviewed, and reviewed again:
2000: FDA conducts in-depth review
2008: review by the Government Accountability Office
2018: review by the Government Accountability Office
All confirmed that the FDA followed its standard practice when it approved the medication.
The question posed in Case #2 — “Is mife is safe and effective?” — is answered by the 23-year track record of mife’s use in the U.S. and safety data documented in more than 100 peer-reviewed articles. As YLE has written before, the scientific evidence resoundingly establishes that medication abortion is safe and effective. Major complications (those requiring hospitalization, surgery, or blood transfusion) following use of mife are extremely rare, occurring in 0.3% of cases. (As a comparison, 1.4-2.3% of women have major complications after childbirth.)
What does this mean today?
Today, people can still get mife and miso for abortion.
Will this change in the future? It’s unclear.
The ball is now in the hands of appellate courts within the U.S. Justice system. The case will likely go to the Supreme Court.
In the meantime, it’s unclear how the FDA and the drug manufacturers will respond, given that the FDA is under contradictory rulings.
The FDA could use “enforcement discretion”—elect not to enforce the Texas ruling for the medications within its purview. This is not the same thing as ignoring the court, but is a long-recognized power of the FDA that the Supreme Court approved of in 1985. They can do this with mifepristone because the Texas judge did not order the FDA to take any particular action—the judge is saying the approval was never valid in the first place. There’s nothing for the FDA to “do.”
Bottom line
Medical choices for pregnancy care are being tested. This is the first time a judge has stayed approval of a drug over the objection of the FDA and the drug manufacturer—a precedent that could upend our medication ecosystem beyond abortion. Regardless, confusion alone can be a strong deterrent to care, as we saw throughout the pandemic.
We should all be closely watching how the appeals play out and how the FDA responds.
Love, YLE, HM, RR, DC
In case you missed previous posts:
Heidi Moseson, PhD, is a reproductive epidemiologist and scientist at Ibis Reproductive Health. She studies abortion access in the U.S. with a particular focus on self-managed abortion with medications.
Rachel Rebouche is a reproductive health scholar and Professor of Law at Temple University.
David S. Cohen is a reproductive rights scholar and Professor of Law at Drexel University.
“Your Local Epidemiologist (YLE)” is written by Dr. Katelyn Jetelina, MPH PhD—an epidemiologist, data scientist, wife, and mom of two little girls. During the day she works at a nonpartisan health policy think tank and is a senior scientific consultant to a number of organizations, including the CDC. At night she writes this newsletter. Her main goal is to “translate” the ever-evolving public health science so that people will be well equipped to make evidence-based decisions. This newsletter is free thanks to the generous support of fellow YLE community members. To support this effort, subscribe below:




.
Moira Donegan
The Guardian
11 Apr 2023
...It doesn't matter if you live on the coasts, or if your governor is Democratic, or if Biden won your congressional district in 2020 by more than 20 points. It doesn’t matter if you live in a city that has more gay bars than churches or where every church flies a pride flag; it doesn’t matter if every mom you know planned all of her pregnancies, and it doesn’t matter if you’ve been managing your own reproduction rights with flexibility and privacy for your whole adult life.
It doesn’t matter if you think you’re safe or that abortion will always be legal where you live. Because the anti-abortion movement wants to impose a national ban on abortion and to take away your right to one. And they already have enough of their partisans in high enough positions – in elected office, yes, but mostly on the federal courts – to do so right now.
That’s the theory behind an injunction that was issued late last Friday by Matthew Kacsmaryk, a Trump-appointed district court judge in Amarillo, Texas, who issued a nationwide injunction invalidating the FDA’s 23-year-old approval of mifepristone, the first of two drugs used in a standard medication abortion. It is the most significant ruling affecting abortion access nationwide since Dobbs.
Mifepristone has been through the most rigorous medical testing possible, has a lower rate of serious complications than Tylenol and has been used to safely and effectively terminate pregnancies by hundreds of thousands of women both in the US and around the world for decades. It will remain available for now, as Kacsmaryk stayed his order for seven days so as to allow the Biden administration time to appeal, which they have done.
Complicating matters further, another district court in Washington state issued a conflicting injunction in another lawsuit – ordering the FDA to keep mifepristone on the market – just an hour after Kacsmaryk’s ban order was issued. The result is a complicated legal struggle, now almost certainly headed to the US supreme court, in which the lives, health and self-determination of millions of American women – and the dignity and full citizenship of all of them – hang in the balance.
Medication abortions using mifepristone now account for more than half of abortions in America. If you’ve ever had an abortion that you only had to take pills for, one of the pills you took was mifepristone. If the ruling goes into effect, abortion providers will be forced to perform only surgical abortions – which are more invasive and demand more resources – or medication abortions using only the second drug, misoprostol – which, while also very effective, are more painful and time-consuming.
There is already a thriving market of mail-order mifepristone in America, much of which is sent into the country from overseas, and which might have in theory evaded Kacsmaryk’s ban; but his ruling also revives the 1873 Comstock Act, a long-dormant and partially invalidated federal statute that prohibits sending abortion drugs or instruments through the mail, while also banning large swaths of material labeled “obscenity”.
Beyond its revival of a Victorian morality law, Kacsmaryk’s injunction is a dramatic escalation in anti-abortion jurisprudence, embracing fringe and extremist positions on abortion, pregnancy and women’s rights, adopting theories of judicial authority and legal standing that range form the tenuous to the bizarre, relying on discredited studies and anti-abortion junk science as statements of fact, and using exclusively the inflammatory, inaccurate and sensationalizing rhetoric of the anti-abortion movement. Throughout, the opinion refers to all pregnant women as “mothers”. Fetuses and embryos are described as “unborn humans”, or “unborn children”.
Kacsmaryk has never been shy about his personal views. For years, he sat on the board of an anti-abortion center, where his sister gave birth at 17; before his appointment to the bench by Trump, he worked at a rightwing legal shop that brought anti-abortion legislation. His injunction order embraces theories long held by only the most extreme fringes of the anti-abortion movement.
He makes empirically untrue claims about the supposed harms of mifepristone specifically, and abortion in general, to women, and he equates reproductive rights to eugenics. He gestures at dark conspiracy theories in his account of the original approval of mifepristone in 2000. And he signals and embrace of fetal personhood, a concept which, if enshrined in law, would ban all abortion nationwide, outlaw many kinds of birth control, curtail women’s access to medical treatments for everything from pre-eclampsia to depression to cancer, and vastly increase the government’s ability to monitor, surveil and punish a long list of activities in the daily lives of pregnant and reproductive-age women.
The order, in short, has none of the traditional judicial pretexts of impartiality or indifference to the policy outcome; it is not a document concerned with the law, but with enforcing an ideology. It reads like the rantings of an extremist activist, because it is.
But because Kacsmaryk is a judge, now those rantings have the force of law. If allowed to stand, Kacsmaryk’s order would deeply curtail access to abortion nationwide. Women in blue states and red states alike would find themselves degraded and humiliated by the law, taken out of control of their own lives, treated by the state as something less worthy than citizens and less competent than adults. But this was always how it was going to go: ever since the supreme court overturned Roe.
The current system, that tenuous balance in which abortion is nominally legal in some states, banned and sadistically punished in others, was never going to hold. The anti-abortion movement was never going to allow women in legal states to remain free; the pro-choice movement, at least the worthy parts of it, was never going to leave women in ban states behind.
The supreme court, controlled now by a group of ravenous rightwing partisans hardly more subtle than Kacsmaryk, is going to try to ban abortion nationwide – if not in this case, then in another one, sooner than you think. And the nation’s pro-choice majority, a newly enraged and motivated group of women voters and their allies, will stand up to them – with more fury than they anticipate.
Moira Donegan is a Guardian US columnist
How is it that all of a sudden, circuit courts are able to issue nationwide "rulings"? Also, how is it that the TX "ruling" applies nationally, but the WA one does not? This doesn't past the smell test.
I have some pretty strong anti Christian views and this issue is a large part of why. If they don't want an abortion, the answer is simple., don't have one. I reject the premise of their religious mythology being used as a basis to dictate people's rights.