Yesterday, the FDA authorized immunocompromised an additional dose of the mRNA COVID19 vaccine. They purposefully kept the language very vague: “solid organ transplant recipients or those who are diagnosed with conditions that are considered to have an equivalent level of immunocompromise."
Then, as per usual, the ball went to ACIP for approval. They were also in charge of defining the “who”, “what”, and “when” for immunocompromised. The ACIP meeting was today. Here are your cliff notes…
Evidence for an additional dose
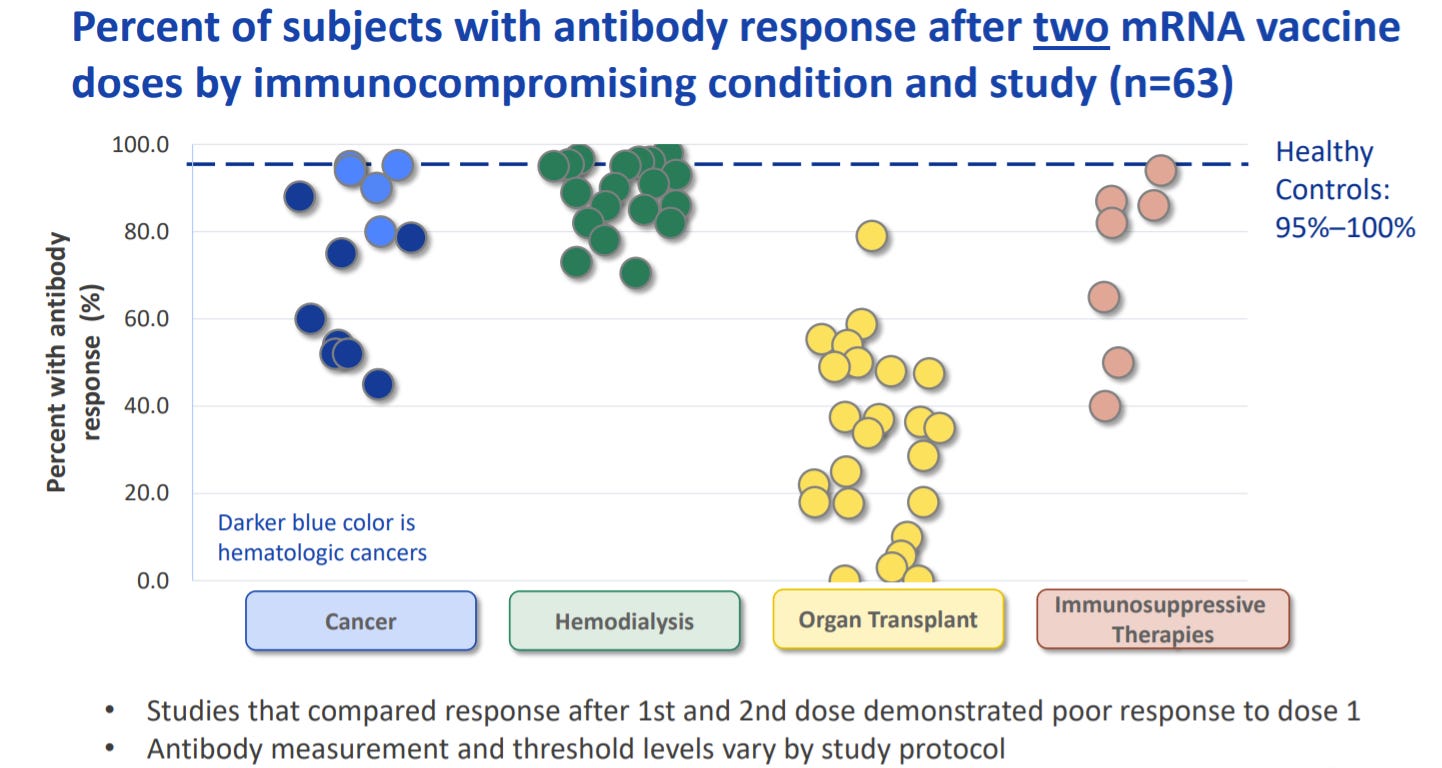
There is overwhelming evidence that this additional dose is needed for immunocompromised. More than 63 studies have shown reduced efficacy of COVID19 vaccine among certain groups, like organ transplants, some cancer patients, and some patients on immunosuppressive medications.
But this decision was really driven by two specific studies:
A randomized control trial with Moderna (here) found immunocompromised patients with a third dose had better protection compared to the placebo (55% vs. 18%)
A French study showed significant improvement after a third dose. No adverse safety events from a third dose occurred.
Other countries have already approved an additional dose for immunocompromised too
France (April 11, 2021) – 3rd dose for patients who are “severely immunocompromised”, but could be extended at a later date to include a larger immunocompromised population
Israel (July 11, 2021) – People living with organ or stem cell transplants, blood cancer, autoimmune disease and treatment with specific immunosuppressive medications – People with breast, lung, or colon cancer did not qualify
UK (July 1, 2021) – Additional dose for immunocompromised people ≥16 years to be implemented in September
Germany (August 2, 2021) – Immunocompromised persons
Who needs to the additional dose?
Unfortunately, there is just not enough evidence for J&J recipients. So people with a J&J vaccine are not authorized for an additional dose.
Among those that got a mRNA vaccine before, these groups need an additional dose:
Active treatment for solid tumor and hematologic malignancies
Receipt of solid-organ transplant and taking immunosuppressive therapy
Receipt of CAR-T-cell or hematopoietic stem cell transplant (within 2 years of transplantation or taking immunosuppression therapy)
Moderate or severe primary immunodeficiency (e.g., DiGeorge, Wiskott-Aldrich syndromes)
Advanced or untreated HIV infection
Active treatment with high-dose corticosteroids (i.e., ≥20mg prednisone or equivalent per day), alkylating agents, antimetabolites, transplant-related immunosuppressive drugs, cancer chemotherapeutic agents classified as severely immunosuppressive, TNF blockers, and other biologic agents that are immunosuppressive or immunomodulatory
What additional dose do immunocompromised need?
The additional dose should be the same mRNA vaccine as the primary series. So, if you got Moderna before, stick to Moderna. You can mix vaccines if your original vaccine is not available.
When do they need the third dose?
The third dose should be administered at least 28 days after second dose.
What else should immunocompromised do?
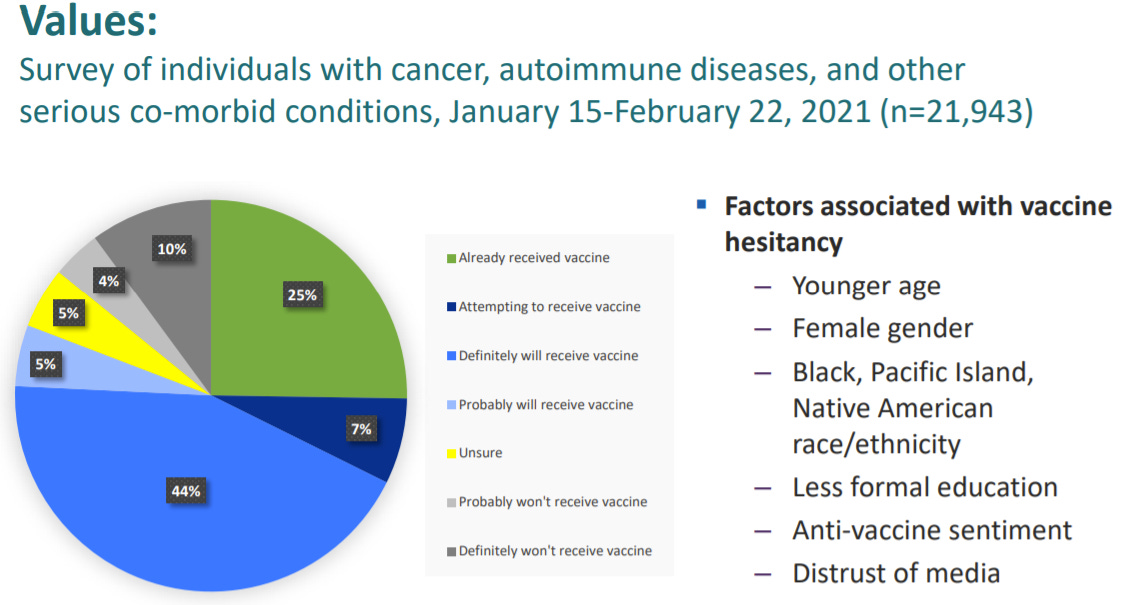
If immunocompromised don’t already have the first two doses, they really need to get going. Although this data is a bit old by now, not nearly enough immunocompromised have even the original vaccine regimen.
Immunocompromised people (including those who receive an additional mRNA dose) should know about the potential for reduced immune response to COVID-19 vaccination
Immunocompromised needs to continue following the usual prevention measures:
Wear a mask
Stay 6 feet apart from others they don’t live with
Avoid crowds and poorly ventilated indoor spaces
Close contacts of immunocompromised people should strongly consider vaccinations, if they haven’t already.
These groups can now go get a third dose. If you ever have specific questions, please reach out to your healthcare provider.
Be safe out there,
YLE





Probably nobody knows the answer to this, but I got both shots, Moderna, and then about a month later started a TNF blocking medication. Does that mean I should get a third shot?
Can you please explain the fear of ADE after the vaccine? The EU chief virologist spoke in a steam about quarantining vaccinated people. This is very disturbing to me.