We got the first round of results from the Moderna booster trial (here is the study and here is the press release).
Booster shots
A few months ago, Moderna started its Phase II trial with booster shots designed against the two similar variants of concern: B1.351 (S. Africa) and P.1 (Brazil). Specifically, they are testing three booster shot strategies:
mRNA-1273.351: I’ll call this 351. This is basically a vaccine that includes instructions made JUST for the S protein found in the B.1.351 variant. This is called a monovalent vaccine- it’s designed to immunize against a single antigen.
mRNA-1273.211: I’ll call this 211. This is a mixed vaccine: 50% with the original vaccine and 50% with 351 booster. This is called a polyvalent vaccine-it’s designed to immunize against two or more strains
mRNA-1273: I’ll call this 1273. This is just a smaller dose (half actually) of the original vaccine that we already got.
This update is on the effectiveness and safety of 351 booster compared to the 1273 booster.
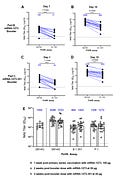
This was a “rollover” study, which means people in the original Phase III Moderna trial were invited to participate in the booster trial. If someone had at least 6 months of follow-up time since their original two-dose series and voiced interest, they were either given 351 or 1273. There were 20 people in each group.
Safety
351 had less side effects:
351: fatigue (37%), headache (37%), muscle pain (32%), fever (0%)
1273: fatigue (70%), headache (55%), joint pain (50%), muscle pain (45%), fever (15%)
Effectiveness
351 also worked better
351: Before the booster, variants had a 7.7 fold decrease in antibody neutralization. This is not surprising, as we know these variants are concerning. After the booster, though, there was a 2.6 fold decrease in neutralization. Which is great!
1273: Before the booster, variants had a 7.3-fold decrease in antibody neutralization. After this booster, there was a 5.3 fold decrease in neutralization. This is better, but not as good as 351.
We’re waiting on more results to see how well 211 works.
We’ll probably have a booster by the end of the year. And probably annually for at least a few years until we get transmission down, so mutations slow down.
By the way, we just got word of the first animal trial on a flu/COVID19 combo vaccine by Novavax. So, ideally and eventually we would only need one shot a year.
Love, YLE


Is the expectation that a booster will only be effective if the original vaccine was from the same manufacturer or at least mRNA vs adenovirus?