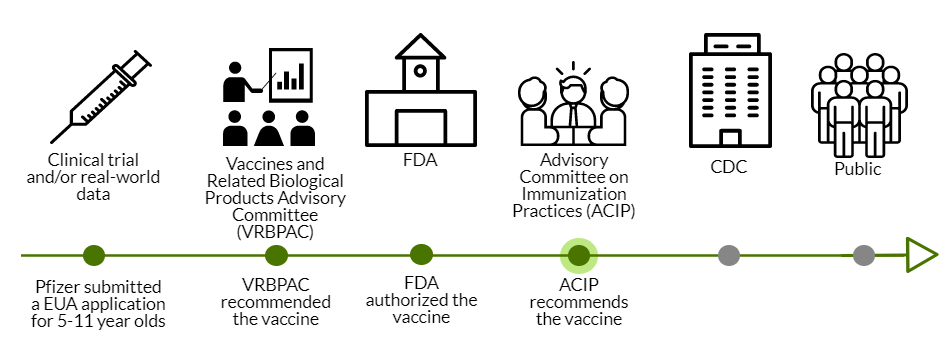
Last week, VRBPAC recommended and FDA authorized the COVID19 vaccine for 5-11 year olds. As per usual, the ball was then passed to ACIP today.
ACIP is an external scientific advisory aboard to the CDC. The 15-person committee compromises a mix of pediatricians, clinicians, researchers, epidemiologists, and professors around the country with no conflicts of interest. Here is the committee roster.
There are also representatives from many national organizations on ACIP including the American Academy of Pediatricians, National Institutes of Health, American Medical Association, etc. While these representatives can’t vote, they are able to discuss, ask questions, and provide perspective.
Today, ACIP needed to accomplish two things:
Determine safety and effectiveness of the vaccine;
Define and clarify the policy: “Who”, “what”, and “when” for 5-11 year olds.
Here was the agenda. Here were the presentations. And here are your cliff notes…
Safety and effectiveness
Overall, ACIP determined the vaccine is safe and effective. I won’t go into detail because much of the same data was presented last week at the VRBPAC meeting. Here are my cliff notes from that meeting.
There was one important detail that was added this week:
Vaccine-induced myocarditis deaths: The CDC presented data on the investigations into vaccine-induced myocarditis deaths among people younger than 30 years old. Among 86 million doses, there have been 9 reports of vaccine-induced myocarditis deaths. Among these 9 cases, 6 have been fully investigated thus far. Three deaths were confirmed as myocarditis. Importantly, all three were due to classic myocarditis (caused from infection of a bacteria/virus) and not due to the vaccine. No myocarditis deaths have been linked to the vaccine in the United States.

Who needs the COVID19 vaccine?
Kids aren’t spared from the harm of COVID19. Briefly, more than 1.9 million have been infected, 8300 have been hospitalized, and 94 have died (making it the #8 killer for 5-11 year olds). Long COVID19, missing school, and driving community transmission are also negative impacts of SARS-CoV-2 among 5-11 year olds. I went over the data in great detail from the VRBPAC meeting.
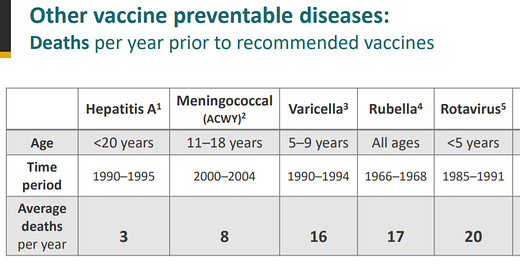
Today, the CDC gave an interesting, and incredibly important, perspective. They compared other vaccine-preventable diseases to COVID19 (see figure below). We vaccinate for diseases that have far less deaths and hospitalizations than COVID19. For example, we vaccinate children for hepatitis A. Hepatitis A had an annual average of 3 deaths before vaccines compared to COVID19 that has an annual average of 66 deaths before vaccines.
Throughout the meeting, evidence was presented that the vaccine would benefit all children aged 5-11 years old.
Children should get the vaccine regardless of underlying health conditions. Among pediatric COVID19 hospitalizations, 32% of children did not have underlying health conditions. Severe COVID19 disease seems to be very random for children.
Children should get the vaccine regardless of previously recovering from COVID19 disease. An estimated 38% of children aged 5–11 years have detectable antibodies from natural infection. This is higher than estimates among adults. But natural immunity, on a population-level, is insufficient in protecting kids. This is due to four reasons:
Naturally immunity protection is random: Some people will mount a strong protection and some people won’t get protection. We have no way to predict who will land in each category (Harvard is working on it). The risk of reinfection after natural infection is 2 - 5 times higher than risk of reinfection with vaccines.
Naturally immunity is protective at first. For some, protection can last 8 months (because we have 8 months of data, it’s probably more). For others, though, protection wanes within weeks. Again, who gets long protection from natural immunity is random.
Vaccination following infection increases protection from subsequent infection, including in the setting of more infectious variants.
Clinical trials in children show that vaccines can be given safely to those with evidence of a prior SARS-CoV-2 infection

Children should get a vaccine even if they have a positive antibody test. This is because of the naturally immunity reasons above and an additional three reasons:
Antibody testing cannot determine when a person was infected. So, we don’t know if, and how, their antibodies are waning.
Antibody tests greatly vary in their ability to detect protection. This is particularly true for those >3 months after infection. People can test positive on a commercial antibody test even after other markers of immunological response, such as neutralizing antibodies, have waned.
At this time, there is no FDA-authorized or approved test that providers or the public can use to reliably determine whether a person is protected from infection.
What vaccine will 5-11 year olds receive?
The 5-11 vaccine is different than for 12+ year olds. It’s a different dosage with a different buffer. The vaccine for 5-11 kids has an orange cap compared to 12+ year olds with a purple cap.
Some more details:
The vaccine dosage is not dependent on a child’s size or weight. As opposed to medications, vaccine dosages are based on the maturity of the immune system. The dosage should be based on the child’s age on the day of vaccination.
Errors: If a 5-11 child accidently receives a 30 µg dose for their first dose, they should receive a single age-appropriate 10 µg dose for their second dose 21 days later and should be considered as having a completed primary series. If a child ages 5–11 years inadvertently receives a 30 µg dose for their second dose, they should be considered has having a completed primary series.
When should 5-11 year olds get the vaccine?
The vaccination should not be delayed. If, for example, an 11 year old has a birthday coming up, they should not wait for a higher dosage. Getting a vaccine as soon as possible is of the upmost importance.
If a child turns from 11 to 12 years of age in between their first and second dose and receives 5–11 years 10 µg (orange cap) for their second dose, they do not need to repeat the dose and this is not considered an error per the EUA.
COVID-19 vaccines may be administered without regard to timing of other vaccines. This includes getting a COVID-19 vaccine with other vaccines on the same day. If multiple vaccines are administered at a single visit, administer each injection in a different injection site.
Discussion
After the presentations, ACIP members openly discussed the safety and effectiveness of vaccines and the policy for universal vaccination of 5-11 year olds. I don’t think I’ve ever heard a stronger, more unified recommendation from all members of ACIP. All members strongly voiced their confidence in the data and the robust view of the situation at the moment. One member said, “this is a huge step forward for children and parents”. Another member said, “we know more than we don’t know”. Even Dr. Bell, who always has a lot of great questions and healthy skepticism, strongly voiced her positive support.
Vote
So, ACIP needed to vote: “The Pfizer-BioNTech COVID-19 vaccine is recommended for children 5-11 years of age in the U.S. population under the FDA’s Emergency Use Authorization”.
Yes: 14 votes
No: 0 votes
The ball now goes to the CDC. If the CDC and FDA agree, then vaccines will officially be authorized and can go into the arms of 5-11 year olds. Unofficially, this may take a few days given distribution difficulties.
COVID19 will now be a vaccine-preventable disease for 5-11 year olds.
Cheers, YLE
For “right away” parents:
When your child gets the vaccine, PLEASE enroll them into V-safe. Here is an overview of V-safe and how to register. You will get the follow-up text messages (not your 5-11 year old).
For “wait and see” parents or “definitely not” parents:
First and foremost, please have a conversation with your child’s pediatrician. They are there to listen and know the health and medical history of your child best.
I joined the President of the American Academy of Pediatrics last week for a Q&A regarding the vaccine for 5-11 year olds. We discussed and addressed many parental concerns. See the recording here.
Here are the top 8 parental concerns of the COVID19 vaccine and my response





Thank you - such great content as always! Now if you could just make my county set up appts in advance ! 😬🤷🏼♀️ We are ready!!
Thank you for this clear and useful information, as always! If at some point you are able to post an update about where we are on vaccines for those younger than 5, this mother of a 4 year old and an 8 month old would love to know!