Today, Merck announced that it’s submitting an FDA application for a new antiviral that can be used against COVID19: molnupiravir. If approved, high risk people would take four capsules twice a day for five days early in their infection. This pill series cuts the risk of hospitalization or death in half. Two other antiviral pills (by Pfizer and Atea Pharmaceuticals and Roche) are expected within the next few months.
As an epidemiologist, we mainly focus on prevention— avoid getting the disease altogether. This is the best case scenario as we avoid morbidity and mortality. And, in the case of a pandemic, reduces transmission and avoids overcrowded health systems. For COVID19, we have an amazingly effective and safe prevention tool: Vaccines.
But even if someone gets the vaccine, they still have a very small chance of ending up in the hospital. Immunocompromised vaccinated people could certainly not be protected too. And there’s of course there’s millions of unvaccinated. So, if someone gets the disease, how can we help them?
Medical Treatment
Vaccines and medical treatments, like antivirals, are incredibly different. But the biggest difference is memory. Vaccines can be given at any time and, if that person is exposed later down the road, it remembers the virus and fights back. Antivirals are only effective when it’s administered within a certain time frame before or after exposure. Then it’s only effective during the time it’s being administered.
There are a few mechanisms in which medical treatment can help fight viruses:
Block the virus from replicating. For example, the FDA approved using Remdesivir for COVID19. This is an antiviral that was originally created for Hep C and has been used against Ebola. For COVID19, it doesn’t save lives, but some evidence shows that it speeds up recovery. Scientists debate on the effectiveness though. So much so that the WHO didn’t recommend using Remdesivir.
Mimic the immune system to help fight an infection. For example, we use monoclonal antibodies. This is a treatment derived from people who have survived COVID19. The problem is that it works best when it’s given early in an infection. By the time someone is hospitalized, it won’t help much.
Temper the immune system from overreacting. For example, we use an anti-inflammatory called dexamethasone when someone’s on a ventilator or receiving oxygen. If it’s given too early, though, it can sometimes do harm.
Other treatments, like ventilators and prone positioning.
Unfortunately, we only have 1 FDA approved treatment for COVID19 (Remdesivir) and 4 additional treatments that are widely used in the United States (monoclonal antibodies, dexamethasone, ventilators, and prone positioning). There are 13 treatments with tentative or mixed evidence. And that’s about it. This is compared to more than 104 vaccines in the pipeline right now.
So, why don’t we have more treatments?
Antivirals are incredibly difficult to create because viruses can change fast (think flu), antivirals are very specific to a certain disease (we usually can’t use an HIV antiviral for COVID19), and/or scientists have very small areas to target for treatment.
A virus’s lifecycle is shown in the figure below. Scientists try and create antivirals to stop each one of these steps. But each step has it’s own challenge. I’ll explain…
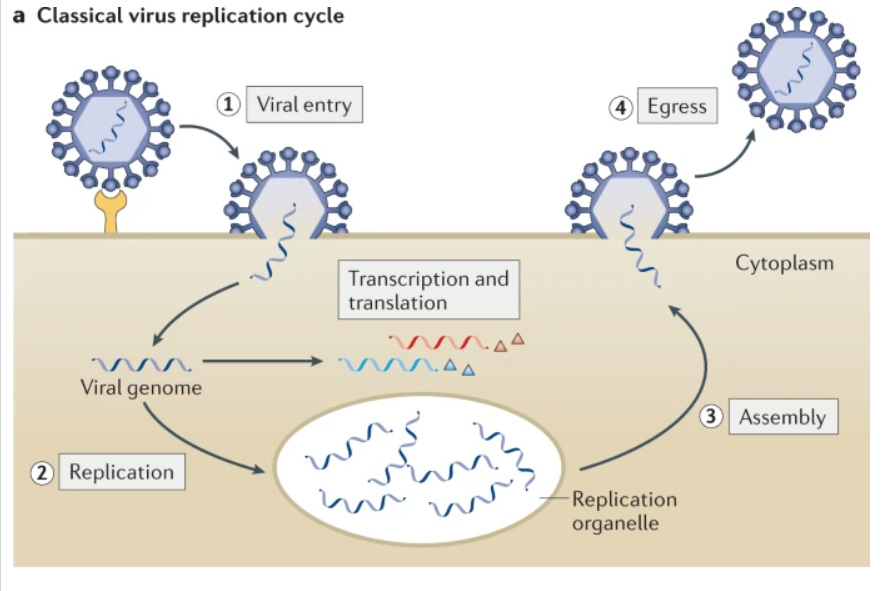
The virus enters the human body and searchers for a host cell. Some scientists try to create antivirals that kill the virus’s protein before it gets to the cell. This has been proven very difficult to do because of the virus’s protective shell. That shell is incredibly durable so it can survive the environment before getting to it’s target.
Viral entry. Then the virus latches to the cell (#1 in the figure) to insert it’s protein. Some scientists try and find a way to stop this step. The challenge is that chemicals that can be used to stop this are toxic for humans.
Replication. Then, scientists try and stop the virus from replicating once in the cell (#2 in the figure). This is challenging because antivirals have to find a way to get to the virus protein without killing the cell altogether. For some diseases (like HPV and HIV), the infected cells “sleep” and stop replicating, which adds a layer of treatment complexity.
Escape. Finally, some scientists try and stop the virus from escaping to go infect other cells. This is how oseltamivir (Tamiflu) helps fight the flu. Tamiflu stops the virus from dissolving its way out and so it can’t go infect others. This helps people recover from the flu 1 or 2 days earlier.
In all of these steps, the antivirals don’t eradicate the virus directly. They just stop (or slow down) viruses from spreading within the body. So it’s important we get the drug to the person as soon as possible. It also means that the immune system is still important.
Bottom Line: Antiviral drugs are incredibly difficult to make and certainly not as effective as vaccines, but I’m excited to see the progression of Merck’s drug. We can use all the help we can get.
Happy Friday, YLE





More excellent info! I don't know how you do it! This was just fresh news today and you have your usual excellent explainer out already.
Ok but where are the under 5 vaccines? This is great, but I feel like no one's really doing anything with those--all I've seen or heard about is the over 5 group and I can't convince any pediatricians to do an off label vaccine. Will I have to wait until my babies are 5 before we can leave the house?