This week I’m diving deep into long COVID. This mini-series was originally 5 posts total, but to be honest I couldn’t keep up. So, I combined #4 and #5. This is the final post of the series.
In epidemiology, one of the biggest jobs we have is to find disease patterns. Because if we can find patterns, the disease is predictable. And if it’s predictable, then it’s preventable and/or treatable. This means we are not only interested in how many people get the disease, but who gets that disease.
For the past two years, scientists have been trying to figure out the patterns of long COVID. So far, we’ve uncovered a few very interesting patterns.
Females
One of the first clues we received was that females reported long COVID far more often than men. A French study on hospitalizations found that between May and July 2020, women outnumbered men 4 to 1 for reporting long COVID. Women who reported long COVID were also much younger compared to those that who didn’t—an average of 40 years old. Since then, dozens more studies across the world have confirmed this finding. Most recently, we saw that young adolescent girls reported long COVID 12 months after infection while adolescent boys did not.
Three hypotheses for this are being explored:
Autoimmunity. We are getting more and more evidence that long COVID is due to autoimmunity (see more below). Autoimmune disorders are far more common in women than men. One study estimated that 80% of all patients diagnosed with an autoimmune disease are female. So, more autoimmunity = more long COVID.
Pregnancy Compensation Hypothesis. This theory suggests that over time younger women (i.e., reproductive age) evolved to have more active immune responses. This evolution allowed more protection against a virus during pregnancy. If SARS-CoV-2 is lingering in viral reservoirs, younger women’s immune systems will be even more heightened. This could result in higher prevalence.
Seeking care. There may also be a behavioral aspect. Before the pandemic, females were far more likely to seek medical care than men in general. So, long COVID may have been underreported by men. But this may only play a small role in long COVID patterns. The Kaiser Family Foundation interestingly found that the pandemic reversed this association—women were less likely to receive preventative care than men.
Comorbidities: diabetes and asthma
We are also uncovering patterns of long COVID with diabetes. Interestingly, the relationship seems to be bi-directional: COVID seems to cause type II diabetes and type II diabetes seems to cause long COVID. For example:
COVID → Diabetes: In a JAMA study that included over 2 million people, the rate of new type 2 diabetes was about twice as high in people who had tested positive for COVID-19 compared with those who had tested negative.
Diabetes → long COVID: In another seminal study, researchers found that those with type II diabetes were more likely to have long COVID 2-3 months after initial infection.
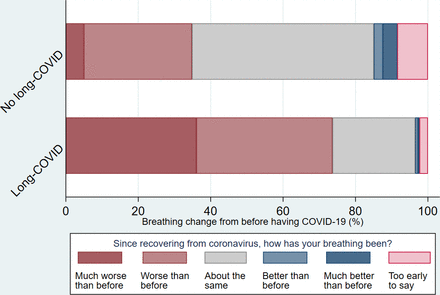
Asthma has also been linked to long COVID. The first study, published in March 2021, found that asthma was the only preexisting condition significantly associated with long COVID after 28 days. Another important study followed this and found that 56% of asthmatic patients reported long COVID, including difficulty breathing (see figure below). The relationship between asthma and long COVID is complex and, as the authors of the second study said, “it is unclear to what extent [prolonged symptoms] are due to one of the multiple pathological processes implicated in long COVID, destabilised asthma, or a combination of factors.”
Viral load
We’ve known for a while that SARS-CoV-2 viral load can predict severe COVID19 disease. One of the leading virology labs, Yale’s Iwasaki Lab, found that viral load on saliva tests predicted the severity of COVID-19 cases, like mortality. This wasn’t too surprising, as we saw this same phenomenon with SARS and MERS.
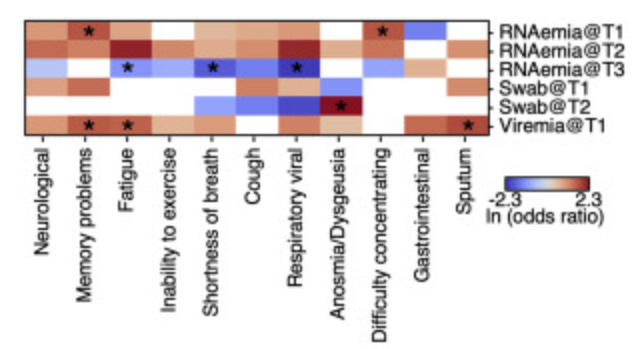
We now know viral load can predict long COVID as well. A recent study found the level of coronavirus RNA in the blood early in infection positively predicts long COVID 3-months later. Interestingly, they found that viral load maps to one specific long COVID symptom: loss of taste and smell.
Presence of EBV and autoantibodies
Autoantibodies are generated as part of the immune system’s response to infection. The problem is that certain autoantibodies can mistakenly start attacking our own body because they see good cells as intruders, when they are not. We’ve seen this with other viruses, like Epstein-Barr virus infection (EBV), which increases the production of autoantibodies and thus may drive multiple sclerosis.
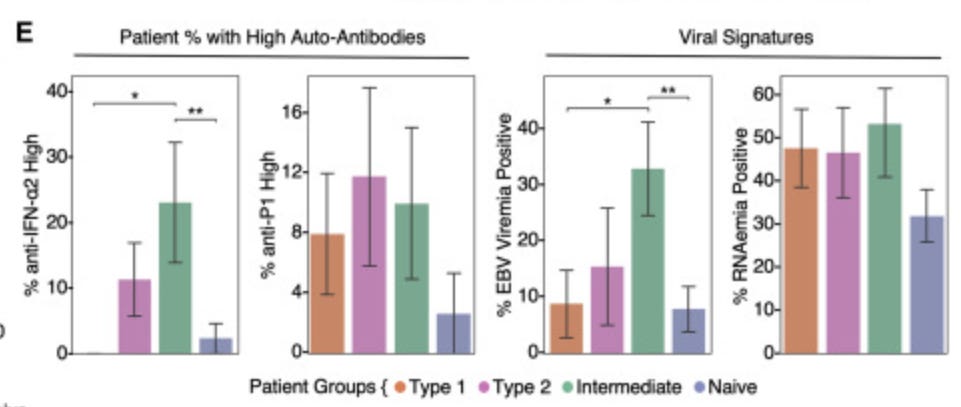
A recent study evaluated whether EBV and autoantibodies are elevated among patients with long COVID. At baseline, 14% of patients had reactivation of EBV. They found that these patients were significantly more likely to have two long COVID symptoms 3 months later: fatigue and productive cough.
They also found that the presence of autoantibodies at infection positively predicted long COVID symptoms for patients. This suggests that patients may be predisposed to long COVID early in infection.
Antibody signature
Another risk factor includes low levels of certain antibodies. In a recent study, scientists evaluated 175 patients with COVID19 and 40 healthy controls and followed their health for up to a year. Those who developed long COVID were found to have lower levels of IgM and IgG3 antibodies.
These scientists then created an algorithm or a long COVID score called an “antibody signature.” This algorithm accurately predicted long COVID in a separate sample of 395 patients. This has incredibly exciting implications because antibody signature could be used by clinicians in the hospital for early identification of patients at very high risk of developing long COVID. Or this antibody signature could be used to inform who needs certain treatments, like immunoglobin.
Treatment
In order to develop new treatments, it’s essential we get a solid grasp on burden, the epidemiological patterns, and the biological pathways through which long COVID develops. As one recent commentary from UCLA outlined:
“We need high-quality data and information that supports an accurate diagnosis before patients can receive appropriate supportive care and effective, disease-specific therapy.”
The patterns just described are starting to provide hypotheses for treatment. For example, will early immunoglobulin treatments help prevent long COVID among those with a particular antibody signature? Or could other therapies help alleviate other COVID symptoms?
Some of these hypotheses have started to be tested. For example, results from one study were presented at the American College of Cardiology’s Cardiovascular Summit conference last month. A scientific group evaluated the impact of enhanced external counterpulsation (EECP) therapy among 50 COVID-19 survivors. EECP uses contracting and relaxing pneumatic cuffs on the calves, thighs, and lower hip area to provide oxygen-rich blood to the heart muscle, brain, and the rest of the body. The sessions lasted for more than 7 weeks. At the end, long COVID symptoms like fatigue, breathing difficulties, and chest discomfort all improved. There was no control group, so this evidence is preliminary but exciting.
While we explore innovative solutions, people need help now. Long COVID is real. We cannot ignore symptoms, like we did with the long history of Chronic Fatigue Syndrome. Long COVID clinics (called Post COVID Care Centers [PCCC]) have opened across the country and across the world. There is a fantastic database of long COVID clinics here (below is a screenshot).
These clinics are approaching care holistically and comprehensively. There won’t be one pill that cures all long COVID patients, as symptoms range too widely, and the pathways that cause these symptoms vary too much. A multi-disciplinary and individualized approach to treatment is key. A fantastic JAMA paper provided an inside look at a Post–COVID-19 clinic and the typical process for patients, which includes a full evaluation and treating the symptoms. For example, one long COVID physician said:
“A good 70% of our patients are experiencing fatigue. We look at their sleep hygiene and try the usual pharmacologic or nonpharmacologic approaches to make sure that people are sleeping well. If there’s depression, anxiety, there are well-validated approaches to treating those symptoms, whether it be pharmacologic or nonpharmacologic.”
Support groups are also important. The website survivorcorps.org is one of the best resources I’ve seen, which describes itself as “connecting, supporting, educating and mobilizing COVID-19 Survivors.” Here and here are other fantastic long COVID websites, specifically for parents of children.
Bottom line
There’s much we don’t know about long COVID but patterns are coming to the surface. These patterns will help develop evidence-based treatments. If you have long COVID, don’t lose hope. Long COVID clinics and support groups can help. And we are getting more answers every day.
This is the conclusion of the mini-series. I hope it opened your eyes to the millions fighting for their health after infection and the many scientists who are working hard to find answers and solutions. And I hope it shows how extensive the SARS-CoV-2 health footprint will be for years to come.
Love, YLE
“Your Local Epidemiologist (YLE)” is written by Dr. Katelyn Jetelina, MPH PhD—an epidemiologist, biostatistician, professor, researcher, wife, and mom of two little girls. During the day she has a research lab and teaches graduate-level courses, but at night she writes this newsletter. Her main goal is to “translate” the ever-evolving public health science so that people will be well equipped to make evidence-based decisions. This newsletter is free thanks to the generous support of fellow YLE community members. To support the effort, please subscribe here:


Thank you so much for all your work in gathering and explaining this complex information to us. I was hoping you were going to address the risks of long covid for fully vaxxed and boosted folks. In my blue bubble, people are very scared to unmask because they are worried about long covid. Can you give us some context and information about what we know about that risk?
Thanks for the very interesting newsletters, it's always great to get a summary of the latest research! I have to point out that one part in this is not correct, however- concerning the pregnancy compensation theory.
>This evolution allowed more protection against a virus during pregnancy.
This is not actually correct, and is wrong also in the Guardian article you linked. The idea behind the pregnancy compensation hypothesis is that during pregnancy, immune reactivity is actually 'dialled down'. The theory says that because the mechanisms that recognize self vs non-self tissues might attack the placenta, which is a mix of parent and offspring tissues, immune reactivity must be lowered.
To compensate for this, immune reactivity is higher during non-pregnant periods- which in a world post-birth control is most of the time for most women. This results in a higher incidence of autoimmune disorders specifically for women. You can see this visualized in the article here: https://pubmed.ncbi.nlm.nih.gov/31200807/#&gid=article-figures&pid=figure-1-key-figure-uid-0
I think it's important to point this out, because the way it is implies that women have more robust immune responses while pregnant, including to SARS-CoV-2- but it's actually more likely to be the opposite. For the story on Long Covid, it's the same result (women have more immune reactivity), but a tiny thing that I think is worth correcting.