Up until now, we had two big unanswered questions regarding COVID19 vaccines in the United States:
Can we safely mix vaccines? For example, if you had an original Johnson and Johnson (J&J) vaccine can you safely get a Moderna booster? We know that mixing AstraZeneca with Pfizer is safe from clinical trials in the UK. Because J&J is the same biotechnology as AstraZeneca, we hypothesized that mixing would be okay too. But needed to see the data.
Can (and how do) we effectively mix vaccines? For example, if you had two doses of Moderna, should you get a third booster of J&J? Is this any better (or worse) than a Moderna booster?
These questions were answered in a landslide study that was just released today. The timing of the preprint is not a coincidence. VRBPAC (external scientific advisory committee to the FDA) is reviewing the evidence for mixing vaccines on Friday. Any data they review and discuss has to be released 48 hours prior to the meeting.
The study
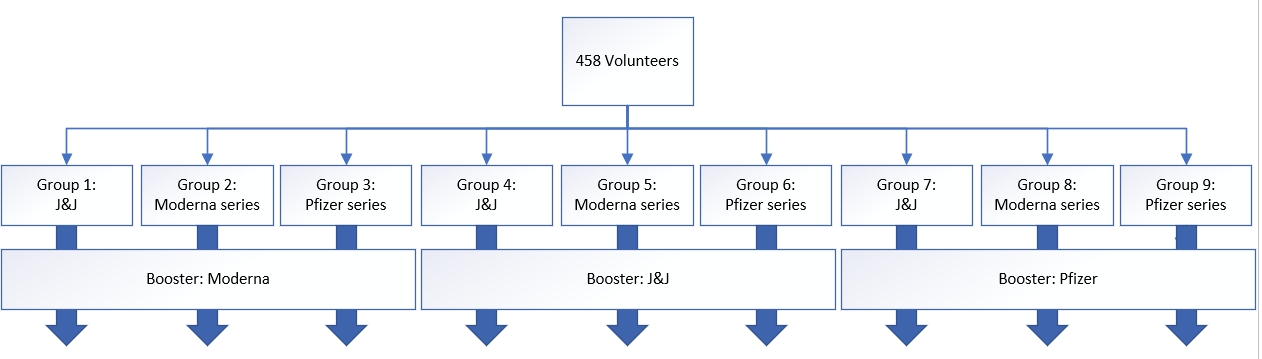
In total, 458 volunteers were divided into nine groups with roughly 50 volunteers in each group. I threw together a figure to show how those people were divided.
Briefly, people that got a primary J&J, Moderna series, or Pfizer series were randomly given a booster four to six months after. For example, those who initially got the two dose Moderna series got either another Moderna shot, a Pfizer shot or a J&J shot.
Then participants were asked to report side effects in the 29 days following. Scientists also drew blood from participants to quantify protection: number of neutralizing antibodies and IgG antibodies. What did they find?
Can we safety mix vaccines?
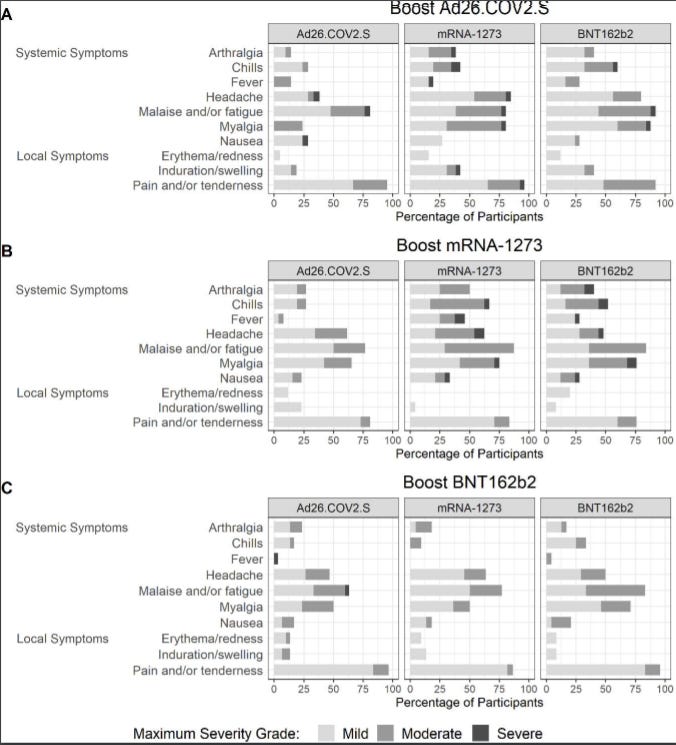
Yes, the safety profile was nothing more than what we expected. Mixing is safe, but you will probably experience a side effect (like chills, fever, headache, or fatigue). In all groups, there were only 2 severe events: renal failure due to a fall injury and gallbladder swelling. After investigation, these events were not linked to the booster. One person reported vomiting 1 day after the J&J booster (Group 5).
Which combination works best? To me, this was the most interesting…
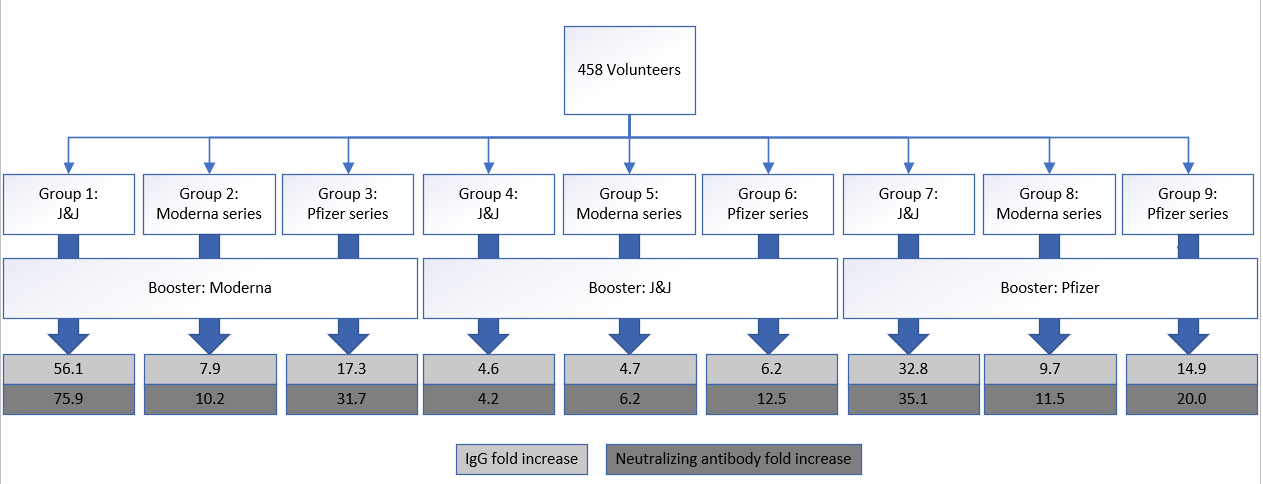
The most benefit, by far, was for those with a primary J&J shot. A Moderna booster gave the greatest benefit to J&J folks (56.1 fold increase in IgG; 76.1 fold increase in neutralizing antibodies). Pfizer also worked very well (32.8 fold increase in IgG; 35 fold increase in neutralizing antibodies). There was minimal increase with a J&J booster (4-fold increase).
After the primary Moderna series, there was benefit from either a Moderna or Pfizer booster. There was really no difference between the two.
After the primary Pfizer series, the greatest benefit was from a Moderna booster. But a Pfizer booster helped a lot too.
A J&J booster following an mRNA series did help, but not as much as another mRNA booster. I thought this was interesting. Scientists originally hypothesized that different vaccine biotechnologies could help more. In theory, your body got more tools to recognize the virus. But that doesn’t seem to be the case. Only higher efficacy matters. This is, importantly, assuming that J&J doesn’t get better with time. In original J&J clinical trials, the most benefit occurred 4 weeks after the original shot. The follow up period for the current study was 29 days, so optimal benefit may have been missed.
Results will be discussed Friday in great detail
This data will be discussed on Friday at the VRBPAC meeting. And, although we finally have some answers it’ll be interesting to see what happens next from a regulatory perspective:
The FDA cannot recommend mixing vaccines because none of the vaccine companies have asked for approval. For example, J&J has not asked to approve mixing with a Moderna booster. So, this will likely be punted to ACIP and the CDC to make a “preferential” recommendation next week. A preferential recommendation doesn’t direct anyone to do anything, but it rather says: “if you do this, this is what the experts recommend and it’s okay”.
Then, the natural next question is can agencies make a preferential recommendation if Moderna is still under an EUA? Or would this have to wait?
Finally, this study included Moderna as a 100 μg booster. The problem is Moderna applied for a 50 μg booster EUA. Would a 50 μg booster change this study’s results? Probably. So… then what?
Other important meetings are coming up too…
And, to make matters more complicated, there are other meetings coming up at the FDA (then the CDC):
Tomorrow: What is the safety, effectiveness, and, most importantly, need of a Moderna (50 μg) booster? There’s not much evidence that Moderna people need a booster so this should be an interesting discussion to follow.
Friday morning: What is the safety, effectiveness, and need of a J&J booster? This should be more straightforward; I expect a booster to be recommended.
Friday afternoon: What is the safety and effectiveness of mixing vaccines?
Oct 26: What is the safety, effectiveness, and need of a vaccine for 5-11 year olds?
Then the FDA will make a decision and then data and decisions go to ACIP and CDC. And THEN there will finally be a formal recommendation. Phew.
We’re starting see a data dump in preparation for all of the scheduled meetings. And, more than ever, we’ll be drinking from a firehose of information. I will (try to) distill the data, meeting information, and cliff notes as we go.
Gear up; this will be a lot.
Love, YLE
And a very big thank you to the 66,066 people that have already filled out my “Who are you” survey! If you haven’t already, please complete it here.




THANK YOU for working so tirelessly! Know that your hard work of researching, communicating, and translating to us is sooo appreciated. You have truly been a lifesaver this year. PLEASE TAKE CARE and go hug your husband and kids now. :)
An anti-vaxxer and an engineer are crossing a bridge over a crocodile-infested river.
The anti-vaxxer asks “What are the odds of us making it across the bridge safely?”
The engineer replies “After a careful structural analysis, I calculate a 99.9% chance of crossing this bridge safely.”
The anti-vaxxer then says “Forget it, I’ll swim.”