
We’re starting to have a pretty good idea of what medications work and don’t work against COVID19 disease. Slowly we are working our way from populist treatment to evidence-based treatment.
Preface: There is a difference between population health (another word for public health and epidemiology) and patient health (another word for clinical care or patient-care). The distinction is important and we shouldn’t use them interchangeably, especially when we talk about drugs. Population health fixes problems for populations at a time. It takes into account everything that impacts health: culture, environment, government, and even genetics. Patient health fixes problems for one person at a time. So, for example, Ivermectin might have worked for your uncle or neighbors mother. It may have even worked for 100 patients for one doctor. But this does NOT mean it works for the rest of the population. This is important to distinguish because population health (not patient health) drives policy. And… this is a blog about population health.
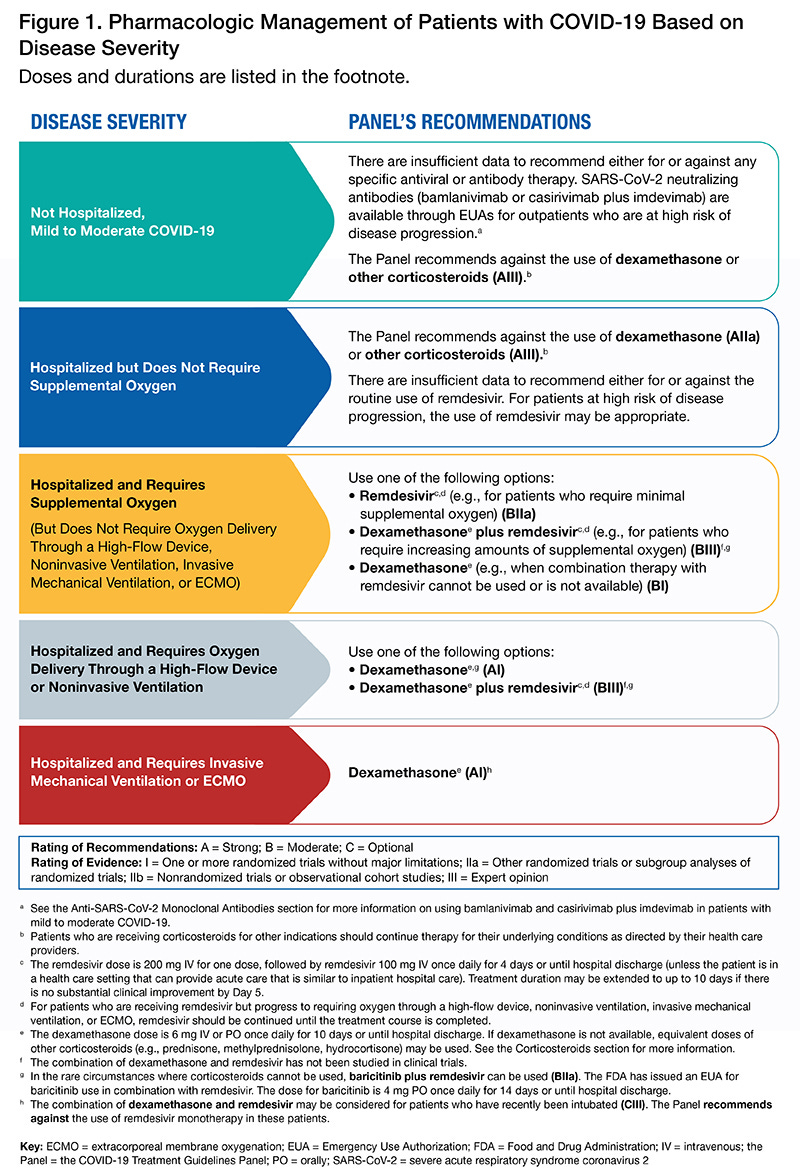
On Feb 11, 2021, the National Institutes of Health published a 280 page report summarizing COVID19 treatment options. Treatment options were based on severity of disease (see Table for summary). And, really, there’re only two options that work so far:
Corticosteroids: This has been consistently effective against death from severe and critical COVID19 disease. Corticosteroids also help prevent mechanical ventilation and decrease ICU length of stay. There has been one study on mild COVID19 disease and corticosteroids did NOT help.
Remdesivir: This is the only FDA-approved antiviral drug found effective for COVID-19. And, specifically, only for people hospitalized needing supplemental oxygen (NOT ventilation).
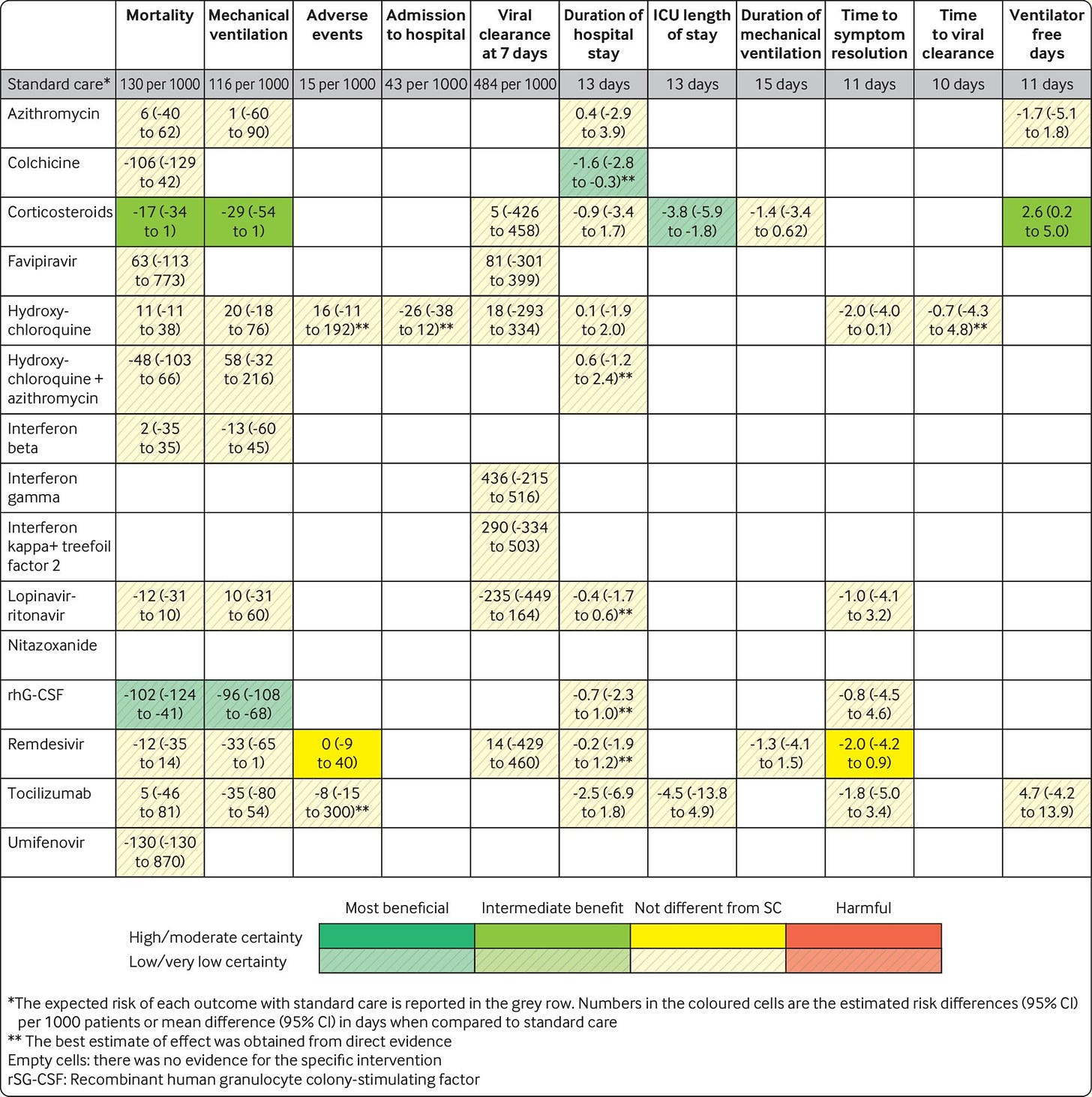
We know a lot of drugs do NOT work against COVID19. In the beginning of the pandemic, we were just throwing spaghetti on the wall to see what stuck. And, well, a lot of it didn’t stick. Here’s a beautiful table summarizing the evidence from the British Medical Journal. It was updated on Dec 10, 2020. Each row corresponds with a different drug. The rows with all yellow boxes (like azithromycin or hydroxychloroquine) mean that these drugs had no impact on COVID19 outcomes (like mortality or ICU stay). In other words, there was no difference in getting hydroxychloroquine compared to a placebo.
Then we have other drugs that are making headlines. Making headlines does NOT mean that we (necessarily) have the scientific evidence needed to recommend treatment for a population. We still need a lot of evidence on:
Ivermectin: Results, thus far, are mixed. We aren’t too surprised they’re mixed, though, because our bodies need a 100-fold increase dose of Ivermectin (than what is currently approved for human use) for antiviral efficacy. Ivermectin, though, does have the potential for anti-inflammatory mechanisms, and thus improving COVID19 outcomes. So, studies persisted. Some studies show no benefits or even worse outcomes among patients given Ivermectin. Other studies have shown that Ivermectin is useful. However, the latter had incomplete information and many limitations, making it difficult to determine if the effectiveness was due to Ivermectin instead of other factors (such as other drugs or demographics). The other problem is that Ivermectin has been linked to tremors, convulsions, lethargy and disorientation. In 2018, scientists found that Ivermectin can pass from the bloodstream to the brain in people with a genetic mutation, ultimately causing brain damage. So, we don’t know if benefits outweigh the risks… “The [NIH] Panel cannot draw definitive conclusions on the clinical efficacy of ivermectin for the treatment of COVID-19”. Merck basically echoed the same thing on Feb 4, 2021.
Tocilizumab (arthritis medication): This drug reduced death by 14% (596 deaths in tocilizumab group and 694 deaths in the placebo group) in one recent study.
Budesonide (asthma medication): Inhaled budesonide reduced the risk of urgent care or hospitalization by 90% when compared to usual care.
That’s it for now!
Love, YLE









Great article as usual, but it's been a little while now. Could you maybe revisit this with any new data that may have come out on Ivermectin and Hydroxychloriquine, as well as maybe touch on the research behind molnupiravir and Paxlovid?
Nice, thank you.